Altafluor Drug Description: Detailed Datasheet
- Generic Name: fluorescein sodium and benoxinate ophthalmic solution
- Brand Name: Altafluor
side effects drug center altafluor (fluorescein sodium and benoxinate ophthalmic solution) drug
Drug Description
ALTAFLUOR
(fluorescein sodium and benoxinate hydrochloride) ophthalmic solution, USP 0.25%/0.4% (Sterile)
For Use in the Eyes Only
DESCRIPTION
Fluorescein Sodium and Benoxinate Hydrochloride Ophthalmic Solution, USP 0.25%/0.4% is a disclosing agent with rapid anesthetic action of short duration.
Fluorescein Sodium is represented by the following structural formula:
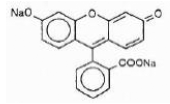 |
Mol. Wt.= 376.27
C20H10Na2O5
Chemical Name: Spiro [isobenzofuran-1 (3H), 9 -[9H] xanthene]-3-one, 3’,6’ dihydroxy, disodium salt.
Benoxinate Hydrochloride is represented by the following structural formula:
 |
Mol. Wt.= 344.88
C17H28N2O3 · HCl
Chemical Name: 2-(Diethylamino) ethyl 4-amino-3butoxybenzoate monohydrochloride
EACH mL CONTAINS: ACTIVES: Fluorescein Sodium 2.5 mg (0.25%), Benoxinate Hydrochloride 4 mg (0.4%); INACTIVES: Povidone, Boric Acid, Water for Injection. Hydrochloric Acid may be added to adjust pH (4.3-5.3). PRESERVATIVE: Chlorobutanol 1%.
Indications & Dosage
INDICATIONS
For procedures requiring a disclosing agent in combination with a topical ophthalmic anesthetic agent such as tonometry, gonioscopy, removal of corneal foreign bodies and other short corneal or conjunctival procedures.
DOSAGE AND ADMINISTRATION
Usual Dosage
Removal of foreign bodies and sutures, and for tonometry, 1 to 2 drops (in single instillations) in each eye before operating.
HOW SUPPLIED
Fluorescein Sodium and Benoxinate Hydrochloride Ophthalmic Solution, USP 0.25%/0.4% is supplied in a glass bottle with a sterilized dropper in the following size: 5 mL.
Storage
Store in a refrigerator at 2°-8°C (36°-46°F), can be stored at room temperature for up to 1 month. Keep tightly closed.
FOR USE IN THE EYES ONLY.
DO NOT USE IF IMPRINTED SEAL ON CAP IS BROKEN OR MISSING.
KEEP OUT OF REACH OF CHILDREN
Manufactured: ALTAIRE PHARMACEUTICALS, INC. Aquebogue, NY 11931 USA. Revised: May 2014
Side Effects & Drug Interactions
SIDE EFFECTS
Occasional temporary stinging, burning and conjunctival redness have been reported after use of ocular anesthetics, as well as a rare, severe, immediate-type, apparent hyper-allergic corneal reaction, with acute, intense and diffuse epithelial keratitis, a gray, ground glass appearance, sloughing of large areas of necrotic epithelium, corneal filaments and sometimes, iritis with descemetitis.
Allergic contact dermatitis with drying and fissuring of the fingertips has been reported.
TO REPORT SUSPECTED ADVERSE REACTIONS, contact Altaire Pharmaceuticals, Inc., at 1-800-258-2471 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
No information provided.
Warnings & Precautions
WARNINGS
Not for Injection. Topical Use Only. Prolonged use of a topical ocular anesthetic is not recommended. It may produce permanent corneal opacification with accompanying visual loss. Avoid contamination-do not touch tip of sterile dropper used to dispense solution to any surface. Replace container closure immediately after using.
PRECAUTIONS
This product should be used cautiously and sparingly in patients with known allergies, cardiac disease, or hyperthyroidism. The long-term toxicity is unknown; prolonged use may possibly delay wound healing. Although exceedingly rare with ophthalmic application of local anesthetics, it should be borne in mind that systemic toxicity manifested by central nervous systems stimulation followed by depression may occur. Protection of the eye from irritation, chemicals, foreign bodies and rubbing during the period of anesthesia is very important. Tonometers soaked in sterilizing or detergent solutions should be thoroughly rinsed with sterile distilled water prior to use. Patients should be advised to avoid touching the eye until the anesthesia has worn off.
Use In Specific Populations
Pregnancy: Pregnancy Category C.
Animal reproductive studies have not been conducted with Fluorescein Sodium and Benoxinate Hydrochloride Ophthalmic Solution USP, 0.25%/0.4%. It is also not known whether Fluorescein Sodium and Benoxinate Hydrochloride Ophthalmic Solution USP, 0.25%/0.4% can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Fluorescein Sodium and Benoxinate Hydrochloride Ophthalmic Solution USP, 0.25%/0.4% should be given to a pregnant woman only if clearly needed.
Nursing Mothers
Caution should be exercised when Fluorescein Sodium and Benoxinate Hydrochloride Ophthalmic Solution USP, 0.25%/0.4% is administered
to a nursing woman.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Overdosage & Contraindications
OVERDOSE
No information provided.
CONTRAINDICATIONS
Known hypersensitivity to any component of this product.
Clinical Pharmacology
This product is the combination of a disclosing agent with a rapidly acting anesthetic of short duration.
Medication Guide
PATIENT INFORMATION
No information provided. Please refer to the WARNINGS and PRECAUTIONS sections.
